
Advancing Drug Discovery Through Cryogenic Science
CryoLabs is a cutting-edge pharmaceutical research laboratory that integrates advanced cryogenic principles and technologies into every stage of drug development and formulation. Our mission is to harness the unique properties of extreme cold to enhance the stability, efficacy, and shelf-life of pharmaceutical products.
Comprehensive Cryogenic Research
Our state-of-the-art facilities are equipped for every stage of pharmaceutical development, leveraging the power of low-temperature science.

Formulation Development
Innovative cryogenic formulation techniques for enhanced drug stability and delivery
• Freeze-drying optimization
• Cryoprotectant development
• Stability enhancement protocols
• Novel delivery systems

Drug Discovery
Advanced cryogenic screening and molecular analysis for novel compound identification
• High-throughput cryogenic screening
• Molecular stability analysis
• Protein crystallization studies
• Compound library preservation

Analytical approach
Comprehensive analytical testing using cutting-edge cryogenic methodologies
• Thermal analysis studies
• Cryogenic microscopy
• Stability testing protocols
• Quality control assays
State-of-the-Art Cryogenic Facilities
Our laboratory features the most advanced cryogenic equipment and methodologies, enabling breakthrough research in pharmaceutical development

Controlled Atmosphere Labs
Precision environmental control

Cryo-Electron Microscopy
Molecular structure analysis

ISO Certified Processes
Quality assurance standards

Ultra-Low Temperature Storage
-196°C liquid nitrogen systems
Our Advanced Facilities & Specifications

Cryogenic Storage Systems
Temperature Range: -196°C to -150°C
Storage Capacity: 10,000+ samples
Vapor Phase Technology: Yes
Auto-Fill System: Integrated
Monitoring: 24/7 remote monitoring
Backup Systems: Redundant cooling
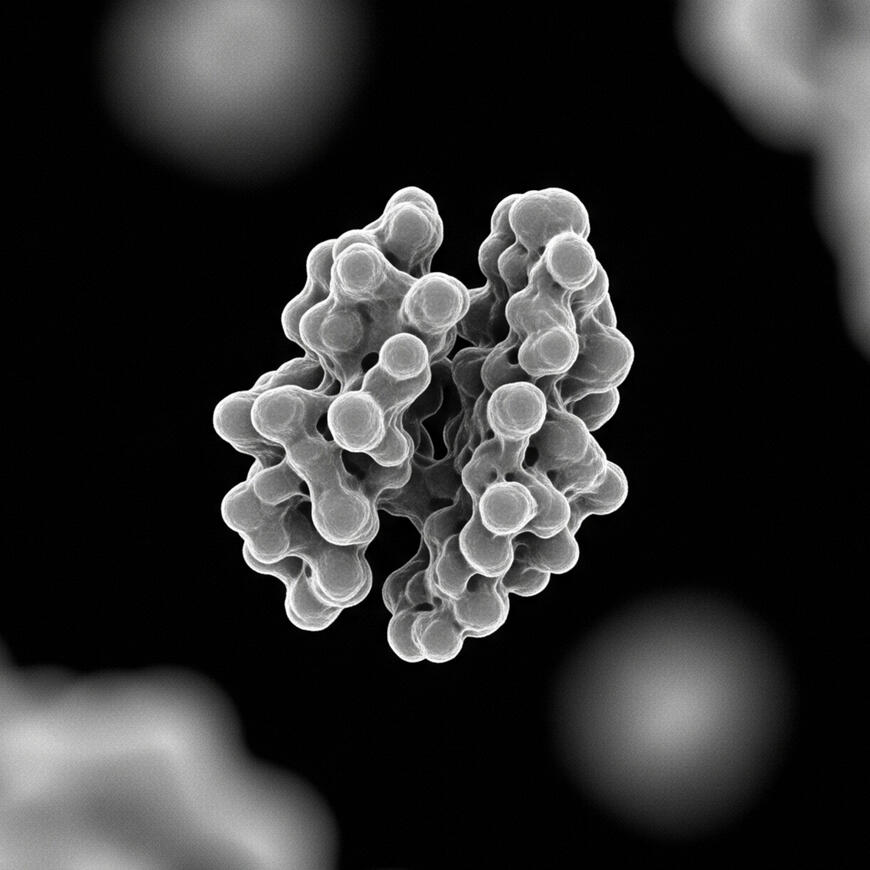
Cryo-Electron Microscopy
Resolution: Sub-angstrom (0.8 Å)
Acceleration Voltage: 300 kV
Magnification: 50x to 2,000,000x
Sample Temperature: -180°C
Direct Detection: K3 Summit Camera
Automation: SerialEM compatible

Freeze-Drying Technology
Shelf Temperature: -55°C to +70°C
Vacuum Level: < 5 mTorr
Shelf Area: 2.3 m² (24.7 ft²)
Ice Capacity: 25 kg/24h
Control System: SCADA w/ 21 CFR Part 11
Validation: IQ/OQ/PQ documented

Clean Room Facilities
Classification: ISO 14644-1 Class 7
Air Changes:n60+ per hour
HEPA Filtration: 99.97% at 0.3 μm
Pressure Differential: +15 Pa minimum
Temperature Control: 20°C ± 2°C
Humidity Control: 45% ± 5% RH

Analytical Equipment
HPLC-MS/MS: Triple Quad 6500+
Detection Limit: pg/mL range
DSC Range: -180°C to +725°C
TGA Sensitivity: 0.1 μg
FTIR Resolution: 0.09 cm⁻¹
Compliance: 21 CFR Part 11

Expert Research Team
PhD Scientists: 15+ researchers
Experience: 10-25 years average
Specializations: Cryobiology, Pharmaceutics
Publications: 200+ peer-reviewed
Patents: 50+ filed/granted
Certifications: GLP, GMP trained
Compliance & Certifications
Quality Standards
• ISO 9001:2015 Certified
• ISO 13485:2016 Medical Devices
• ISO 14644 Clean Room Standards
Regulatory Compliance
• FDA 21 CFR Part 11
• GLP/GMP Guidelines
• ICH Q1A-Q1E Stability
Safety & Security
• OSHA Safety Protocols
• Biosafety Level 2 Certified
• 24/7 Security Monitoring
Our Misson
Pioneering the Future of Pharmaceutical Research
At CryoLabs, we believe that the integration of cryogenic science with pharmaceutical research holds the key to unlocking new therapeutic possibilities. Our team of world-class scientists and researchers are dedicated to pushing the boundaries of what's possible in drug discovery and development.
15+ years of specialized cryogenic research experience
Partnerships with leading pharmaceutical companies
50+ patents in cryogenic pharmaceutical applications
Published research in top-tier scientific journals
Ready to Advance Your Research?
Partner with CryoLabs to leverage cutting-edge cryogenic science for your pharmaceutical research and development needs.
Get In Touch
Ready to explore how cryogenic science can advance your pharmaceutical research? Contact our team of experts today.
Email Us
Get in touch with our research team[email protected]
Call Us
Speak directly with our experts+34 936 941 269
Lun-Vie, 9:00-18:00 CET
Visit Us
Tour our state-of-the-art facilitiesParc Científic, 08028 Barcelona

© 2025 CryoLabs. All rights reserved. Advancing pharmaceutical research through cryogenic science.